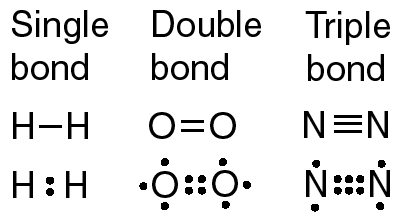Key Points
-bond- a force that holds groups of two or more atoms together and makes them functional as a unit
-bond energy- the energy needed for breaking a bond
-ionic bonds- the attraction between a strongly positive ion and a strongly negative ion
-covalent bond- a bond is formed between atoms when electrons are shared by the nuclei
-ionic compound- a compound that is formed by an ionic bond [for example, a metal (+) and a nonmetal (-)]
-polar covalent bond- a bond in which a pair of electrons is shared in common between two atoms, but the pair is held more closely by
one of the atoms, making it unequal
-electronegativity- the relative ability of an atom in a molecule to attract a shared electron to itself
-dipole moment- the product of magnitude of charge on a molecule and the distance between charges of equal magnitude with opposite
signs
In almost all stable chemical compounds of representative elements, all of the atoms have achieved a noble gas electron configuration
-ionic compounds tend to form small crystals
-ionic compounds are solid at room temperature
-ionic compounds have low thermal conductivity
-bond energy- the energy needed for breaking a bond
-ionic bonds- the attraction between a strongly positive ion and a strongly negative ion
-covalent bond- a bond is formed between atoms when electrons are shared by the nuclei
-ionic compound- a compound that is formed by an ionic bond [for example, a metal (+) and a nonmetal (-)]
-polar covalent bond- a bond in which a pair of electrons is shared in common between two atoms, but the pair is held more closely by
one of the atoms, making it unequal
-electronegativity- the relative ability of an atom in a molecule to attract a shared electron to itself
-dipole moment- the product of magnitude of charge on a molecule and the distance between charges of equal magnitude with opposite
signs
In almost all stable chemical compounds of representative elements, all of the atoms have achieved a noble gas electron configuration
-ionic compounds tend to form small crystals
-ionic compounds are solid at room temperature
-ionic compounds have low thermal conductivity
Lewis Structure
A representation of a molecule that shows
Valence electrons
-electrons on the outer shell
Representative metals form ions by losing enough electrons to achieve the configuration of the previous noble gas
Nonmetals form ions by gaining enough electrons to form the configuration of the next noble gas
-a single bond involves two atoms sharing one electron pair
-a double bond involves two atoms sharing two pairs of electrons
-a triple bond involves two atoms sharing three electron pairs
-electrons on the outer shell
Representative metals form ions by losing enough electrons to achieve the configuration of the previous noble gas
Nonmetals form ions by gaining enough electrons to form the configuration of the next noble gas
-a single bond involves two atoms sharing one electron pair
-a double bond involves two atoms sharing two pairs of electrons
-a triple bond involves two atoms sharing three electron pairs
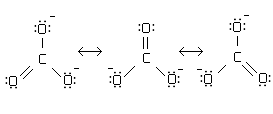
Resonance
-a molecule shows resonance when more than one Lewis can be drawn for the molecule
-a molecule shows resonance when more than one Lewis can be drawn for the molecule
Example Calculation
1. What is bond energy?
2. What is a covalent bond?
3. Label each of these bonds as covalent, polar covalent, or ionic:
a) K--Cl
b) Br--Cl
c) Cl--Cl
4. Give the total number of valence electrons in each of the following compounds
a) B2O3
b) CO2
c) CBr4
d) C2H2
5. Draw a Lewis dot structure for the following:
a) C2H6
b) CHCl3
c) C2H4Cl2
d) N2H4
2. What is a covalent bond?
3. Label each of these bonds as covalent, polar covalent, or ionic:
a) K--Cl
b) Br--Cl
c) Cl--Cl
4. Give the total number of valence electrons in each of the following compounds
a) B2O3
b) CO2
c) CBr4
d) C2H2
5. Draw a Lewis dot structure for the following:
a) C2H6
b) CHCl3
c) C2H4Cl2
d) N2H4
Answers
1. Bond energy is the energy required to break a chemical bond
2. A covalent bond exhibits the sharing go electrons between nuclei
3. a) ionic b) polar covalent c) covalent
4. a) 24 b) 16 c) 20 d) 17
5. :
2. A covalent bond exhibits the sharing go electrons between nuclei
3. a) ionic b) polar covalent c) covalent
4. a) 24 b) 16 c) 20 d) 17
5. :
Resources
http://quizlet.com/15699263/chemistry-ch-12-flash-cards/
http://hyperphysics.phy-astr.gsu.edu/hbase/chemical/bond.html
http://www.watchknowlearn.org/Video.aspx?VideoID=36997&CategoryID=5993
http://www.schooltube.com/video/7870b1153b034ec08d7a/
http://hyperphysics.phy-astr.gsu.edu/hbase/chemical/bond.html
http://www.watchknowlearn.org/Video.aspx?VideoID=36997&CategoryID=5993
http://www.schooltube.com/video/7870b1153b034ec08d7a/